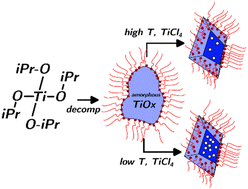
Partially diminished types of titanium dioxide (generally referred to as “black” titania) have attracted widespread curiosity as promising photocatalysts of oxidation on account of their absorption within the seen area. The principle approaches to supply it depend on postprocessing at excessive temperatures (as much as 800 °C) and excessive pressures (as much as 40 bar) or on extremely reactive precursors (e.g., TiH2), and yield powders with poorly managed sizes, shapes, defect concentrations and distributions. We describe an method for the one-step synthesis of TiO2 colloidal nanocrystals at atmospheric stress and temperatures as little as 280 °C. The temperature of the response permits the density of oxygen vacancies to be managed by almost two orders of magnitude independently of their dimension, form, or colloidal stability. This artificial pathway seems to supply vacancies which are homogeneously distributed within the nanocrystals, somewhat than being concentrated in an amorphous shell. Because of this, the defects are protected against oxidation and lead to steady optical properties in oxidizing environments.