|
Take heed to this text |

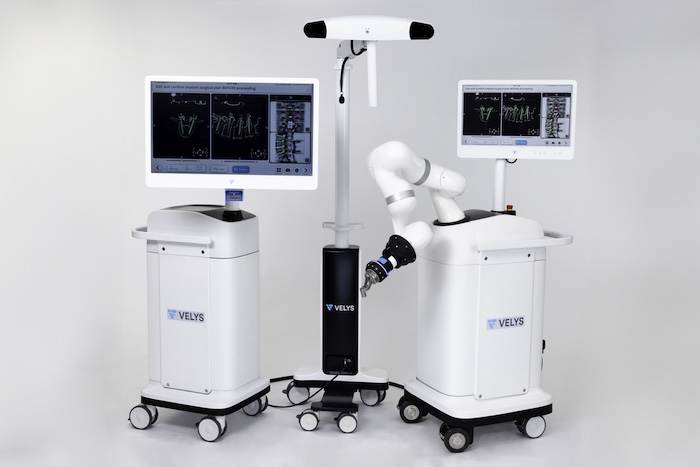
Velys Backbone, a system for planning and instrumenting spinal fusion procedures. | Credit score: J&J/DePuy Synthes
Johnson & Johnson’s DePuy Synthes this week launched a dual-use robotics and standalone navigation platform that makes use of its Velys robotic in backbone surgical procedure. This marks the newest growth for Velys, J&J’s robotic orthopedic surgical procedure system that gained its first FDA clearance for whole knee surgical procedure in 2021. In June, Velys gained FDA clearance for unicompartmental knee arthroplasty.
The medtech large’s orthopedics unit developed the backbone surgical procedure Velys system in collaboration with eCential Robotics. eCential introduced FDA 510(okay) clearance for this platform final month. The businesses designed the Velys lively robotic-assisted system (Velys Backbone) for planning and instrumenting spinal fusion procedures. Areas embrace the cervical, thoracolumbar and sacroiliac backbone.
DePuy Synthes mentioned the businesses goal for the know-how to assist surgeons sort out their most advanced challenges. The twin-use system provides standalone navigation and an lively robotics platform for flexibility in method and planning. Lively robotics permits surgical steerage tailor-made to surgeon desire.
The corporate mentioned the distinctive options and capabilities of lively robotics might “set up a brand new normal in backbone surgical care.”
Velys Backbone provides a customizable expertise with pathology-specific workflows, aided by choices just like the Velys adaptive monitoring know-how and Velys trajectory help. It joins the DePuy Synthes core backbone portfolio of merchandise, the corporate mentioned.
DePuy Synthes — the most important orthopedic machine firm on the earth — anticipates full industrial availability within the first half of 2025.
“We’re shaping the subsequent frontier of orthopaedic innovation with a relentless concentrate on digital developments and excellence within the subject of surgical robotics and navigation,” mentioned DePuy Synthes chair Aldo Denti. “Our dedication extends to enhancing affected person care by way of important strides in backbone surgical procedure. It is a main step in rising our Velyx Portfolio and in our dedication to supporting backbone surgeons and their sufferers with superior instruments.”
In the meantime, competitor Stryker — which has seen an excessive amount of success with its Mako robotic surgical procedure programs — expects a Mako Backbone launch within the second half of 2024, with Mako Shoulder out by the tip of 2024.